Predominant Collagen Fiber Orientation (CFO) and Osteon Morphotypes
Predominant CFO is often strongly strain-mode specific , 1. (see footnote) and regional CFO patterns are often produced by the regional prevalence of specific osteon morphotypes. In order to quantify these relationships, osteon morphotype scoring, first introduced by Martin and co-workers (1996a), has become a useful method for discerning mechanically relevant variations in the regional distributions of secondary osteons when viewed under circularly polarized light (CPL) (Skedros et al., 2011a, Skedros et al., 2011b).
In a microscopic analysis using CPL and thin plane-parallel transverse sections, the seminal study by Martin et al. (1996a) described six phenotypic variants of the secondary osteons in the diaphyseal cortices of adult equine third metacarpals. In addition to identifying these six variants (osteon ‘morphotypes’), they introduced a numerical method for scoring regional variations in their distribution. Under CPL these osteon morphotypes are distinguished by variations in birefringent (“brightness”) patterns that are attributed to their lamellar collagen organization/orientation. These range from: (1) “hoop” osteons, containing a bright peripheral ring of highly oblique-to-transverse collagen fibers that surround longitudinal fibers, which appear darker in CPL, to (2) “distributed” osteons with highly oblique-to-transverse collagen patterns distributed across the entire osteon wall, which appears bright in CPL (Fig. 1). Martin and co-workers’ scoring scheme, which can be expressed as an osteon “morphotype score” (MTS), represents a significant advance in interpreting bone adaptation because the regional prevalence of these osteon morphotypes appears to be common, having been observed in various bones of diverse species (Beraudi et al., 2010, Skedros et al., 2009, Warshaw, 2007, McFarlin et al., 2008, Skedros et al., 1996, Currey, 2002, Riggs et al., 1993a, Vincentelli, 1978, Skedros et al., 2011b)

Fig. 1 The six-point scoring scheme with examples of each osteon birefringence pattern (osteon “morphotype”). These images are reproduced from the original study of Martin et al. (1996b). The numerical values of each of the six morphotypes are used to calculate the osteon morphotype score (MTS) of entire microscopic images that contain many osteons (Skedros et al. 2009). Four of the six numerical scores shown include consideration of the completeness and birefringence strength (brightness) of the peripheral ring “O” or hoop:
0 = category N, a dark osteon with no birefringent lamellae;
1 = category OWI, a combination of OI and OW;
2 = category OW, similar to O but the birefringent ring is weak (W);
3 = category OI, similar to O but the birefringent peripheral ring is incomplete (I);
4 = category O osteon with dark interior and strongly birefringent peripheral lamellae;
5 = category D, birefringent lamellae are distributed throughout the wall of the osteon (“distributed” osteon group). This group includes “bright” osteons (less frequent) and “alternating” osteons (more frequent).
(Images are reproduced from Martin et al. (1996a) with permission of Elsevier Science, Inc.)
As shown in figure 1, we modified the osteon MTS method of Martin et al. (1996a) as a means for more accurately interpreting relationships of morphotypes with specific load environments (Skedros et al., 2009, Skedros et al., 2011a, Skedros et al., 2011b). In bones that receive habitual bending, studies using CPL images have shown osteon morphotypes with relatively greater amounts of transverse collagen (bright gray levels) distributed in the osteon wall represent adaptation for predominant compression, while osteon morphotypes with more longitudinal collagen (dark) represent adaptation
Footnote 1. “Strain-mode specific refers to adaptations that are strongly correlated with prevalent/predominant (i.e., “habitual”) tension, compression, or shear. For example, predominantly longitudinal CFO tends to occur in habitual tension-loaded regions (“tension regions”) and predominantly oblique-to-transverse CFO tends to occur in habitual compression-loaded regions (“compression regions”)(Figs. 2 and 3). for predominant tension (Figs. 2 and 3) (Riggs et al., 1993a, Riggs et al., 1993b, Skedros et al., 2006a, Skedros et al., 2004, Skedros et al., 2007). Consequently, these differences help identify habitual bending because morphotypes found in tension-loaded regions differ significantly from those in compression-loaded regions. Osteons that appear to be “hybrids” of those in tension and compression regions tend to predominate in the neutral axis regions, probably representing adaptations for prevalent/predominant shear. In contrast to bending, bones or bone regions loaded in prevalent/predominant torsion do not exhibit regional variations in osteon morphotypes — regional variations in osteon morphotypes are not expected here because the relatively more uniform bone matrix organization is believed to represent adaptation for diffusely distributed shear stresses that are produced in this loading mode.
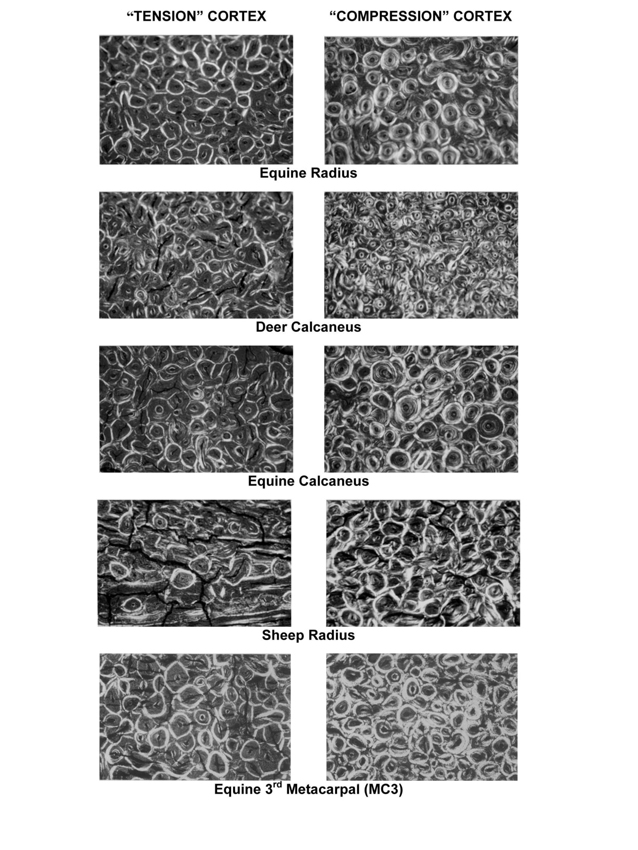
Fig. 2 Representative circularly polarized light (CPL) images of bone from “tension” cortices (at left) and “compression” cortices (at right) of several non-human species (from Skedros et al., 2009). The width of each image is 1.52mm. The specimens were embedded in polymethyl methacrylate in an unstained and fully calcified state, and ultramilled to 100 microns thickness. All images were taken under the same illumination and 50x magnification.

Fig. 3 Representative circularly polarized-light images taken from the proximal diaphyseal cross-sections of an adult chimpanzee femur. At left, are images from the lateral “tension” cortex that shows generally darker osteons, which are quantified as lower osteon morphotype scores using our scoring scheme (e.g., values lower than “5” in Fig. 1). At right, are images from the medial “compression” cortex that shows generally brighter osteons, which are quantified as higher osteon morphotype scores using our scoring scheme (e.g., “5” in Fig. 1). The width of each image is 1.4mm. The specimens were embedded in polymethyl methacrylate in an unstained and fully calcified state, and ultramilled to 100 microns thickness. All images were obtained under the same illumination and magnification.
Early observations and studies of osteon morphotypes
The genesis of studies showing differential mechanical properties of osteon morphotypes can be traced to those of Ascenzi and Bonucci (1967, 1968). These investigators elucidated a correlation between the mechanical properties of individual secondary osteons and the birefringence patterns that they identified: predominantly bright, predominantly dark, or alternating bright and dark lamellae. They observed that the dark osteons had greater strength in tension than those having bright or “alternating lamellae”. Following the interpretation of lamellar structure postulated by von Ebner (1887) and Gebhardt (1906), osteons with alternating lamellae, as seen when viewed between crossed polarizers, were assumed to have plies in which the collagen fibers alternated between longitudinal and transverse directions (Fig. 4). Osteons appearing entirely bright or dark were assumed to have mostly transversely or longitudinally oriented fibers, respectively (Bromage et al., 2003). (See Marotti (1996), Burr and Turner (1998), Yamamoto et al. (2000), and Ascenzi and co-workers (2003, 2006) for discussions regarding alternative explanations that have been considered, but are not as well supported, for the physical bases of these differences in lamellar birefringence). A brief review of observations of osteon morphotypes in polarized light reported from the 1930s through the 1980s can be found in Skedros et al. (1996).
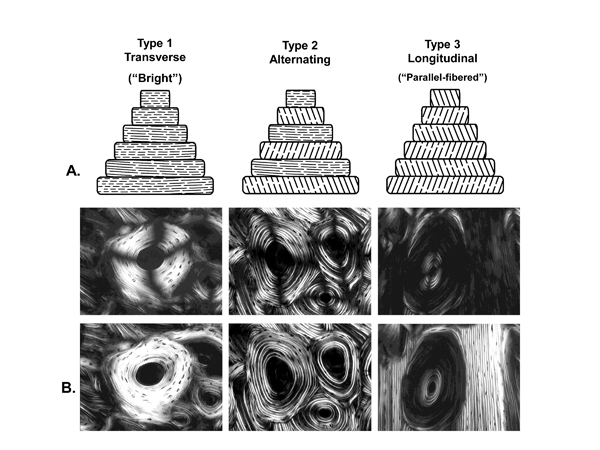
Fig. 4 Examples of three osteon morphotypes in linear (top microscopic images) and circularly polarized light (bottom images; the dark cross-shaped extinction patterns are absent). The illustrations at the top of this figure are diagrammatic depictions of the predominant collagen fiber orientation (CFO) patterns that result in the presence or absence of the birefringence patterns seen in the lamellae of the osteons in the microscopic images. The osteon images are from Bromage et al. (2003), and the layout of the figure is based both on Bromage et al. (2003) and Ascenzi and Bonucci (1968). These investigators ascribe to the view that the birefringence (gray-level) variations, seen for example in the “alternating” osteon, are primarily based on lamellar variations in predominant CFO. Other investigators have argued that variations in collagen fiber density account for the gray-level variations in the alternating osteons (Marotti 1996); this view is not well supported by experimental data and also appears to be influenced by limitations of specimen preparation techniques (Yamamoto et al. 2000).
(Images are reproduced with permission of The Anatomical Record Part B, The New Anatomist; John Wiley and Sons Inc., Malden, MA, USA.)
Strength enhancement’ vs. ‘toughening’.
Conventional wisdom that ‘strength enhancement’ is the goal of regionally predominant CFO and specific osteon morphotypes has been trumped by more recent data showing that ‘toughening’ is the aim. The earlier conventional paradigm stemmed from studies of the relationship between CFO and mechanical properties of macroscopic cortical bone specimens without respect to regional habitual loads. In the contexts of nonspecific or unspecified load histories, studies of cortical bone anisotropy using plane-polarized light (see Bromage et al. (2003) for discussion of how this differs from circularly polarized light) have shown that predominant CFO is correlated with strength or failure strain of tested specimens (Evans, 1958, Vincentelli and Evans, 1971, Evans and Vincentelli, 1974, Reilly and Burstein, 1974, Reilly and Burstein, 1975, Ascenzi, 1988). Martin and Ishida (1989) also quantified birefringence with plane-polarized light and reported that longitudinal CFO was an important predictor of tensile strength in bovine cortical bone. Martin and Boardman (1993) found similar results for the bending strength and elastic modulus of bovine cortical bone. Using four-point-bending to test cortical beams cut from equine radial diaphyses, and both CPL and plane-polarized light analyses of tested specimens, Martin et al. (1996b) showed that longitudinally oriented collagen correlated with greater modulus and monotonic strength. In sum, these data reflect conventional wisdom that the in vivo role for variations in predominant CFO is primarily for affecting strength-related material properties.
By contrast, this conventional paradigm is challenged by more recent studies that have used strain-mode-specific loading (i.e., compression testing of bone habitually loaded in compression, and tension testing of bone habitually loaded in tension). These studies have shown that predominant CFO more strongly influences regional material toughness (energy absorption). For example, in a study of mid-diaphyseal cortical specimens of mature equine third metacarpals, we have examined the relative contributions of multiple material characteristics (CFO, porosity, ash fraction, osteon population density, percent osteon area, and individual osteon cross-sectional area) in strain-mode-specific testing (Skedros et al., 2006a). In tension testing (dumbbell specimens), CFO was one of the top three characteristics in explaining variability in yield stress, ultimate stress, plastic energy absorption, and total energy absorption. In compression testing (cube specimens), CFO was among the top three characteristics in explaining yield energy absorption, plastic energy absorption, and total energy absorption. These results add to a growing body of experimental data that reveal an important role for predominant CFO, collagen content, and/or intermolecular collagen cross-links in affecting energy absorption (a measure of toughness) (Banse et al., 2002, Skedros et al., 2006c, Burr, 2002, Wang et al., 1998, Nyman et al., 2007). In a study of strain-mode-specific mechanical testing of an equine radius (12-year-old Thoroughbred), Reilly and Currey (1999) showed that the patterns of microdamage formed, and the mode of failure of specimens from the anterior “tension” and posterior “compression” cortices, suggest that each cortex was adapted to its respective habitual loading mode (pg. 551):
In comparison to these studies addressing the mechanical importance of regional variations in averaged CFO values, there are only two studies that specifically evaluate the mechanical consequences of differences in the distributions of specific osteon morphotypes. In the first study, Hiller et al. (2003) showed that variations in the distributions of “distributed” (i.e., “bright” and “alternating”) and “hooped” osteons (Fig. 1) in dorsal (anterior), medial, and lateral cortices of equine third metacarpals can affect regional fatigue life and toughness by differentially dissipating energy (via osteonal pullout) (Fig. 5). In mechanical testing studies of equine third metacarpals, we have also implied that regional variations in the prevalence of these various osteon morphotypes produce the regional differences in CFO that strongly influenced energy absorption in strain-mode-specific testing (Skedros et al., 2006a).
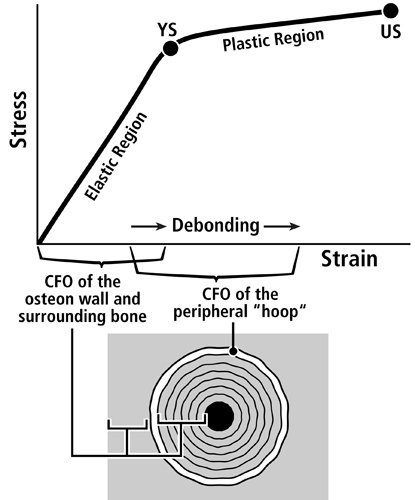
Fig. 5 A. Stress-strain curve of a tension test that shows the hypothesis that the longitudinal collagen that forms the majority of the wall in “tension” osteons helps to satisfy the material requirements of the bone-mineral composite for elastic behavior by curbing excessive strains in order to avoid irreversible damage. The oblique-to-transverse collagen fiber orientation (CFO) of the peripheral lamellae (“hoops”) of these osteons (see Fig. 1) allow for debonding at the cement line in a quasi controlled fashion as the yield point (YS) is approached, and this can continue beyond this point into the initial portion of the plastic region (between YS and US). This “beneficial” debonding dissipates energy that helps to avoid reaching the US point where fracture then occurs. This depiction is based on data and observations described by Bigley et al. (2006), Martin et al. (1998) and Williams et al. (2010).
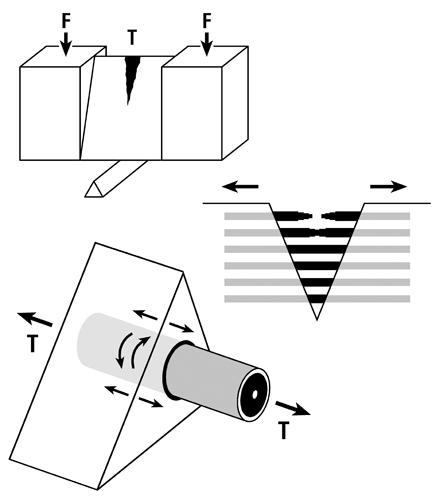
Fig. 5 B. Three drawings depicting an energy absorption (toughness) test in tension (T) of a machined specimen (at top) loaded at each end with a force (F). Energy is absorbed as the osteons debond, pullout, and bridge the forming crack (middle drawing). Osteon morphotypes that appear to be adapted to accommodate habitual tension and resist tension overload have a peripheral lamella with highly oblique/transverse collagen (“hoop”) that probably also absorbs energy by resisting shear stresses (curved lines in lower drawing) during debonding and pull out. (Adapted from Martin et al. (1998)).
In the second study, Bigley et al. (2006) advanced the mechanical pushout methods pioneered by Ascenzi and co-workers and reported important details of the mechanical properties of individual osteons in isolation and with consideration of their surrounding matrix histomorphology. Using the diaphysis of the third metacarpal of an adult thoroughbred horse and observations in both polarized light and scanning electron microscopy, their experimental study revealed clear differences in interfacial strength (at cement line and interlamellar “interfaces”) associated with variations in CFO that typify the four osteon morphotypes that they evaluated (Fig. 6). In their pushout tests, ‘interfacial’ failure was typically observed at the cement line ‘interface’ or within the osteon wall (i.e., interlamellar interface). For alternating osteons the cement line is weaker (12.4 MPa) than the interlamellar interface (33.3 MPa, p = 0.04). By contrast, the cement line is stronger for the bright osteon (47.1 MPa) than the interlamellar interface (33.4 MPa, p = 0.04). Overall, bright osteons had the highest interfacial debonding strength (40.3 MPa), and dark osteons had the lowest strength (22.8 MPa) (p < 0.05). In terms of maximum interfacial shear stress, the bright osteons also had highest stress (82.6 MPa), the dark osteons had the least (63.6 MPa) (p < 0.05), and alternating and hooped osteons had intermediate values (76.3 MPa and 71.0 MPa, respectively). Their linear regression analyses also suggest that there may be a relationship between the amount of oblique-to-transverse collagen fibers (compression-adapted collagen in the osteon wall) and these important mechanical properties, independent of the presence of alternating rings. The histomorphology of the surrounding matrix also profoundly influenced the shear strength of the osteons. For example, as the fractions of bright, dark, and hooped osteons increased, the debonding shear strength increased, whereas when alternating osteons increased, the shear debonding strength decreased. In view of these mechanical test results, it is clear that osteon MTSs that are based in mechanical and morphological contexts should distinguish bright from alternating osteons in addition to distinguishing hooped from non-hooped osteons (Skedros et al., 2011a). In summary, these studies show the mechanical importance of osteon morphotypes.
When strain data are lacking, what is the most reliable structural or material characteristic for interpreting load history in limb bones?

Table 1 lists the main structural and material characteristics to utilize in load history analysis. Among these, consider that the data that are considered in the other sections of this website support the conclusions that predominant CFO and/or osteon morphotypes are the strongest characteristics for interpreting load history — much stronger than mineral content, osteon and osteocyte population densities, osteon shape, and osteon size. In nearly all bones that have been studied, osteon morphotypes produce regional variations in CFO (Skedros et al., 2009, Beckstrom et al., 2010, Skedros et al., 1996, Skedros et al., 2004, Skedros et al., 2007, Mason et al., 1995, Skedros and Hunt, 2004, Skedros and Kuo, 1999, Skedros et al., 2011b). Refer to these papers for data that support CFO as the most important characteristic in this context. There might be a role for mineral crystallite orientation for interpreting load history in cases where preferred alignment of the mineral component (i.e., the extra-fibrillar mineral) of the collagen-mineral composite might be dissociated from the predominant CFO (Skedros et al., 2006b). But techniques required for the less destructive analysis of mineral orientation as an indicator of predominant load direction are not readily available (Espinoza Orias et al., 2009). Consequently, preparing bone specimens for the analysis of patterns in predominant CFO using circularly polarized light is probably the single most important technical step for interpreting the load history of most bones, even if they do not exhibit osteonal remodeling (Riggs et al., 1990, Skedros et al., 2004, Skedros et al., 1996).
Some readers may have selected variations in osteon population density as the strongest characteristic for interpreting load history. In fact, osteon population densities and other osteon morphologic characteristics (e.g., size, shape, etc.) receive great attention in anthropological studies that are aimed at distinguishing species or deciphering the load history of limb bones and ribs (Abbott et al., 1996, Pfeiffer et al., 2006, Mulhern and Ubelaker, 2009, Robling and Stout, 2003, Martiniaková et al., 2006, Urbanová and Novotný, 2005, Burr et al., 1990, Schaffler et al., 1985, Dominguez and Crowder, 2012, Crescimanno and Stout, 2012). In contrast to empirical studies showing strong correlations of CFO with strain-related parameters of load history (Skedros et al., 2006b, Takano et al., 1999), these studies typically show much weaker correlations of osteon populations and other osteon characteristics (e.g., size and shape) with load history and other related mechanical issues. Consequently, these osteon parameters are a distant second to the distribution of osteon morphotypes and/or regional patterns of predominant CFO. Nevertheless, osteon densities in bone must be considered in most cases because they tend to be concentrated (although not always (Gibson et al., 2006, Skedros et al., 2009, Mason et al., 1995)) in the more highly strained regions (“compression” cortices) of bones that are loaded in habitual bending.
In a study of 87 (1 outlier) modern human femoral mid-diaphyseal specimens (45 male, 43 female; 17-97 years; 12,690 osteons), Britz et al. (2009) found an inverse relationship between osteon size and body weight — a relation that was superimposed on the more dominant pattern of decreasing osteon size with age. Although there was no evidence that this relationship was related to strain magnitude, a prior study reported smaller osteons in the femora and tibiae of late Pleistocene adult hominids that was interpreted as reflecting increased strains produced by increased strenuous activity (Abbott et al., 1996). This idea is supported by the analytical study by (van Oers et al., 2008) that showed an inverse relationship between osteon size and strain magnitude in compression (smaller osteons in higher compression). These investigators felt that this relationship mirrored data reported in the literature, especially noting our studies showing smaller osteons in the “compression” cortices (where strains are highest) of the habitually bent calcanei of adult deer, sheep, elk, and horses (Skedros et al., 1994, Skedros et al., 1997). However, this relationship has not been shown in other bones where an association with spatial high-low strain magnitude distributions and osteon size should have been obvious (Mason et al., 1995, Skedros et al., 2009). Additionally, a mathematical error was detected in the Abbott et al. (1996) study mentioned above and a re-examination of their data by Streeter et al. (2010) found that the osteon population densities were not higher and the osteons are not smaller for the presumably more strenuously loaded Pleistocene hominid limb bones. In these perspectives, osteon size is unreliable for interpreting load history. Although there is evidence that osteon size tends to increase toward the endosteum (Britz et al., 2009), inconsistency in this relationship suggests that, when present, it might be an effect of proximity to the marrow and not a consequence of low strain (Skedros et al., 2001, Skedros et al., 1997).
Differences in the distributions of osteocyte densities also caught our attention because of the possibility that this characteristic might strongly correlate with load history, especially in distinguishing bending from torsion. We postulated this relationship because it seemed reasonable that bone tissue could somehow perceive regional non-uniformities in the strain environment (e.g., strain modes and magnitudes). In other words, this “perception” would require the presence of a cellular network that communicates across broad regions of a bone (e.g., up and down a diaphysis or across an entire diaphyseal cross-section). For this to work, the most likely resident cells with this function would be osteocytes because they: (1) are the most numerous bone cells, (2) exhibit modified inter-cellular gap junctions and, hence, cell-to-cell molecular and/or electrical transmission, and (3) form gap junctions with bone lining cells that reside on the porous surfaces where osteoclast migration or activation commences for modeling/remodeling processes (Skedros et al., 2005, Martin et al., 1998, Marotti, 1996). Experimental data show that apoptotic osteocytes, caused by microdamage-induced injury that can occur naturally, trigger the remodeling process (i.e., apoptotic osteocytes attract osteoclasts) (Verborgt et al., 2000, Herman et al., 2010). In these perspectives it seemed reasonable to hypothesize that regional differences in osteocyte densities would be present and would reflect differences in the sensitivity of the cellular network for monitoring and ensuring a normal range of strains in a habitual load environment. However, correlations of osteocyte population density with strain history (modes and magnitudes) and marrow proximity have not been shown in bones where these relationships should be obvious (Skedros et al., 2005, Skedros, 2005). These results further support the conclusion that predominant CFO is the strongest characteristic for predicting load history in these contexts.

Fig. 6 Examples of the osteon morphotypes that were analyzed in pushout tests by Bigley et al. (2006).
(Images are reproduced with permission granted by Lanny Griffin and Elsevier Ltd., Oxford, UK.)
Exceptions to the rule and important caveats.
As expected in biological studies, there are always exceptions to the rule. For example, although strain-mode-related variations in the patterns of CFO can be present in bones where remodeling does not occur (Mason et al., 1995, Skedros and Hunt, 2004), there can be circumstances of growth and/or cases in some bones that poorly remodel where it is difficult or impossible to use this characteristic for deciphering load history. In this perspective, it is important to point out exceptional cases or situations where predominant CFO can be misleading. We have shown the lack of expected strain-mode-specific (tension vs. compression) CFO patterns in Rhesus macaque ulnae and chicken tarsometatarsals even though they are considered to be habitually bent bones (Skedros et al., 2003, Judex et al., 1997, Demes et al., 2001). In the case of the macaque ulnae, the histology in the “tension” and “compression” regions did not show significant differences in CFO and appeared to be comprised of fairly recently deposited laminar bone without much remodeling. By contrast, the lack of expected regional tension vs. compression CFO patterns in the chicken tarsometatarsi evades a similarly straight-forward maturation-related explanation (these bones did show localized osteon remodeling). Thus it may be difficult, or impossible, to discern load history in terms of “expected” histomorphological correlates of functional adaptation in some bones that are growing, have recently stopped growing, and/or are from species that do not exhibit much remodeling (Skedros et al., 2004, Skedros et al., 2007). Examples of this might also be reflected in the seemingly patchy/irregular distribution of several primary histological ‘types’ shown by McFarlin et al. (2008) in various primate limb bones spanning a range from immature to adult. But this does not necessarily mean an absence of correlation between these ‘types’ of primary histology and their overall/regional predominant non-osteon CFO, which the authors did not measure. In turn, many of the bones in their study are also in the “high load complexity” category where regional CFO variations (whether from the primary or secondary bone) would not be expected (see section on “Load-Complexity Categories”).
In summary, these examples show that regional strain-mode-related variations in predominant CFO and/or associated osteon morphotypes, although seem highly reliable for distinguishing habitual bending from torsion in many cases, fall short of being 100% sensitive and 100% specific in this context. This emphasizes an important practical point — investigators must always examine many structural and material characteristics when attempting to interpret the load history of a bone or bone region when in vivo strain data are lacking or insufficient. Perhaps histocompositional characteristics that have been scarcely studied could help in these exceptional cases (e.g., distributions of types and amounts of collagen molecular cross-links or non-collagenous proteins).
References
Abbott S, Trinkaus E, Burr DB (1996) Dynamic bone remodeling in later Pleistocene fossil hominids. Am J Phys Anthropol, 99, 585-601.
Ascenzi A (1988) The micromechanics versus the macromechanics of cortical bone–a comprehensive presentation. J Biomech Eng, 110, 357-63.
Ascenzi A, Bonucci E (1967) The tensile properties of single osteons. Anat Rec, 158, 375-86.
Ascenzi A, Bonucci E (1968) The compressive properties of single osteons. Anat Rec, 161, 377-91.
Ascenzi MG, Ascenzi A, Benvenuti A, Burghammer M, Panzavolta S, Bigi A (2003) Structural differences between “dark” and “bright” isolated human osteonic lamellae. J Struct Biol, 141, 22-33.
Ascenzi MG, Lomovtsev A (2006) Collagen orientation patterns in human secondary osteons, quantified in the radial direction by confocal microscopy. J Struct Biol, 153, 14-30.
Banse X, Sims TJ, Bailey AJ (2002) Mechanical properties of adult vertebral cancellous bone: correlation with collagen intermolecular cross-links. J Bone Miner Res, 17, 1621-8.
Barth HD, Launey ME, Macdowell AA, Ager JW, 3rd, Ritchie RO (2010) On the effect of X-ray irradiation on the deformation and fracture behavior of human cortical bone. Bone, 46, 1475-85.
Beckstrom A, Skedros J, Kiser C, Keenan K (2010) Predominant collagen fiber orientation data support the multi-domain load hypothesis in the chimpanzee femur. Am J Phys Anthropol, Supplement 50, 63.
Beraudi A, Stea S, Bordini B, Baleani M, Viceconti M (2010) Osteon classification in human fibular shaft by circularly polarized light. Cells Tissues Organs, 191, 260-8.
Bigley RF, Griffin LV, Christensen L, Vandenbosch R (2006) Osteonal interfacial strength and histomorphometry of equine cortical bone. J Biomech, 39, 1629-1640.
Britz HM, Thomas CD, Clement JG, Cooper DM (2009) The relation of femoral osteon geometry to age, sex, height and weight. Bone, 45, 77-83.
Bromage TG, Goldman HM, McFarlin SC, Warshaw J, Boyde A, Riggs CM (2003) Circularly polarized light standards for investigations of collagen fiber orientation in bone. Anat Rec, 274B, 157-68.
Burr DB (2002) The contribution of the organic matrix to bone’s material properties. Bone, 31, 8-11.
Burr DB, Ruff CB, Thompson DD (1990) Patterns of skeletal histologic change through time: Comparison of an archaic native American population with modern populations. The Anat. Rec., 226, 307-313.
Burr DB, Turner CH, Naick P, et al. (1998) Does microdamage accumulation affect the mechanical properties of bone? J. Biomech., 31, 337-347.
Crescimanno A, Stout SD (2012) Differentiating fragmented human and nonhuman long bone using osteon circularity. J Forensic Sci, 57, 287-94.
Currey JD (2002) Bones: Structure and Mechanics, Princeton University Press, Princeton, NJ.
Demes B, Qin YX, Stern JT, Jr., Larson SG, Rubin CT (2001) Patterns of strain in the macaque tibia during functional activity. Am J Phys Anthropol, 116, 257-65.
Dominguez VM, Crowder CM (2012) The utility of osteon shape and circularity for differentiating human and nonhuman
Haversian bone. Am J Phys Anthropol, Suppl. 54, 84-91.
Espinoza Orias AA, Deuerling JM, Landrigan MD, Renaud JE, Roeder RK (2009) Anatomic variation in the elastic anisotropy of cortical bone tissue in the human femur. J Mech Behav Biomed Mater, 2, 255-63.
Evans FG (1958) Relations between the microscopic structure and tensile strength of human bone. Acta Anat (Basel), 35, 285-301.
Evans FG, Vincentelli R (1974) Relations of the compressive properties of human cortical bone to histological structure and calcification. J Biomech, 7, 1-10.
Gebhardt W (1906) Über funktionell wichtige anordnungsweisen der feineren und görberen bauelemente des wirbeltierknochens. II. Spezieller teil. Der bau der Haversschen lamellensysteme und seine funktionelle bedeutung. Arch Entw Mech Org, 20, 187-322.
Gibson VA, Stover SM, Gibeling JC, Hazelwood SJ, Martin RB (2006) Osteonal effects on elastic modulus and fatigue life in equine bone. J Biomech, 39, 217-25.
Herman BC, Cardoso L, Majeska RJ, Jepsen KJ, Schaffler MB (2010) Activation of bone remodeling after fatigue: differential response to linear microcracks and diffuse damage. Bone, 47, 766-72.
Hiller LP, Stover SM, Gibson VA, et al. (2003) Osteon pullout in the equine third metacarpal bone: Effects of ex vivo fatigue. J Orthop Res, 21, 481-488.
Judex S, Gross TS, Zernicke RF (1997) Strain gradients correlate with sites of exercise-induced bone-forming surfaces in adult skeleton. J Bone Miner Res, 12, 1737-1745.
Marotti G (1996) The structure of bone tissues and the cellular control of their deposition. Ital J Anat Embryol, 101, 25-79.
Martin RB, Boardman DL (1993) The effects of collagen fiber orientation, porosity, density, and mineralization on bovine cortical bone bending properties. J Biomech, 26, 1047-1054.
Martin RB, Burr DB, Sharkey NA (1998) Skeletal Tissue Mechanics, Springer-Verlag, New York, NY.
Martin RB, Gibson VA, Stover SM, Gibeling JC, Griffin LV (1996a) Osteon structure in the equine third metacarpus. Bone, 19, 165-71.
Martin RB, Gibson VA, Stover SM, Gibeling JC, Griffin LV (1996b) Osteonal structure in the equine third metacarpus. Bone, 19, 165-71.
Martin RB, Ishida J (1989) The relative effects of collagen fiber orientation, porosity, density, and mineralization on bone strength. J Biomech, 22, 419-26.
Martin RB, Mathews PV, Lau ST, Gibson VA, Stover SM (1996c) Collagen fiber organization is related to mechanical properties and remodeling in equine bone. A comparison of two methods. J Biomech, 29, 1515-1521.
Martiniaková M, Grosskopf B, Omelka R, Vondraková M, Bauerová M (2006) Differences among species in compact bone tissue microstructure of mammalian skeleton: use of a discriminant function analysis for species identification. J Forensic Sci, 51, 1235-9.
Mason MW, Skedros JG, Bloebaum RD (1995) Evidence of strain-mode-related cortical adaptation in the diaphysis of the horse radius. Bone, 17, 229-237.
McFarlin SC, Terranova CJ, Zihlman AL, Enlow DH, Bromage TG (2008) Regional variability in secondary remodeling within long bone cortices of catarrhine primates: the influence of bone growth history. J Anat, 213, 308-24.
Mulhern DM, Ubelaker DH (2009) Bone microstructure in juvenile chimpanzees. Am J Phys Anthropol, 140, 368-75.
Nyman JS, Roy A, Tyler JH, Acuna RL, Gayle HJ, Wang X (2007) Age-related factors affecting the postyield energy dissipation of human cortical bone. J Orthop Res, 25, 646-55.
Pfeiffer S, Crowder C, Harrington L, Brown M (2006) Secondary osteons and haversian canal dimensions as behavioral indicators. Am J Phys Anthropol, 131, 460-468.
Reilly DT, Burstein AH (1974) The mechanical properties of cortical bone. J Bone Joint Surg, 56-A, 1001-1022.
Reilly DT, Burstein AH (1975) The elastic and ultimate properties of compact bone tissue. J Biomech, 8, 393-405.
Reilly GC, Currey JD (1999) The development of microcracking and failure in bone depends on the loading mode to which it is adapted. J Exp Biol, 202, 543-552.
Riggs CM, Evans GP, Shah D, et al. (1990) Functional associations in cortical bone between collagen orientation, mechanical properties and in vivo strain history. In 7th Meeting of the European Society of Biomechanics), pp. B5. Aarhus, Denmark.
Riggs CM, Lanyon LE, Boyde A (1993a) Functional associations between collagen fibre orientation and locomotor strain direction in cortical bone of the equine radius. Anat Embryol, 187, 231-238.
Riggs CM, Vaughan LE, Boyde A, Lanyon LE (1993b) Mechanical implications of collagen fibre orientation in cortical bone of the equine radius. Anat Embryol, 187, 239-248.
Robling AG, Stout SD (2003) Histomorphology, Geometry, and Mechanical Loading in Past Populations. In Bone Loss and Osteoporosis: An Anthropological Perspective (eds Agarwal SC, Stout SD), pp. 189-205. New York: Kluwer Academic/Plenum Publishers.
Schaffler MB, Burr DB, Jungers WL, Ruff CB (1985) Structural and mechanical indicators of limb specialization in primates. Folia Primatol, Basel, 61-75.
Skedros JG (2005) Osteocyte lacuna population densities in sheep, elk and horse calcanei. Cells Tissues Organs, 181, 23-37.
Skedros JG, Dayton MR, Sybrowsky CL, Bloebaum RD, Bachus K (2006a) The influence of collagen fiber orientation and other histocompositional characteristics on the mechanical properties of equine cortical bone. J Exp Biol, 209, 3025-3042.
Skedros JG, Demes B, Judex S (2003) Limitations in the use of predominant collagen fiber orientation for inferring loading history in cortical bone. Am J Phys Anthropol, Suppl 36, 193.
Skedros JG, Grunander TR, Hamrick MW (2005) Spatial distribution of osteocyte lacunae in equine radii and third metacarpals: considerations for cellular communication, microdamage detection and metabolism. Cells, Tissues, Organs, 180, 215-236.
Skedros JG, Hunt KJ (2004) Does the degree of laminarity mediate site-specific differences in collagen fiber orientation in primary bone? An evaluation in the turkey ulna diaphysis. J. Anat., 205, 121-134.
Skedros JG, Hunt KJ, Bloebaum RD (2004) Relationships of loading history and structural and material characteristics of bone: Development of the mule deer calcaneus. J Morphol, 259, 281-307.
Skedros JG, Kiser CJ, Keenan KE, Samuel CT (2011a) Analysis of osteon morphotype scoring schemes for interpreting load history: evaluation in the chimpanzee femur. J Anat, 218:480-499.
Skedros JG, Kiser CJ, Mendenhall SD (2011b) A weighted osteon morphotype score out-performs regional osteon percent prevalence calculations for interpreting cortical bone adaptation. Am J Phys Anthropol, 144, 41-50.
Skedros JG, Kuo TY (1999) Ontogenetic changes in regional collagen fiber orientation suggest a role for variant strain stimuli in cortical bone construction. J. Bone Miner. Res., 14, S441.
Skedros JG, Mason MW, Bloebaum RD (1994) Differences in osteonal micromorphology between tensile and compressive cortices of a bending skeletal system: Indications of potential strain-specific differences in bone microstructure. Anat Rec, 239, 405-413.
Skedros JG, Mason MW, Bloebaum RD (2001) Modeling and remodeling in a developing artiodactyl calcaneus: A model for evaluating Frost’s mechanostat hypothesis and its corollaries. Anat Rec, 263, 167-185.
Skedros JG, Mason MW, Nelson MC, Bloebaum RD (1996) Evidence of structural and material adaptation to specific strain features in cortical bone. Anat Rec, 246, 47-63.
Skedros JG, Mendenhall SD, Kiser CJ, Winet H (2009) Interpreting cortical bone adaptation and load history by quantifying osteon morphotypes in circularly polarized light images. Bone, 44, 392-403.
Skedros JG, Sorenson SM, Hunt KJ, Holyoak JD (2007) Ontogenetic structural and material variations in ovine calcanei: a model for interpreting bone adaptation. Anat Rec, 290, 284-300.
Skedros JG, Sorenson SM, Takano Y, Turner CH (2006b) Dissociation of mineral and collagen orientations may differentially adapt compact bone for regional loading environments: Results from acoustic velocity measurements in deer calcanei. Bone, 39, 143-151.
Skedros JG, Su SC, Bloebaum RD (1997) Biomechanical implications of mineral content and microstructural variations in cortical bone of horse, elk, and sheep calcanei. Anat Rec, 249, 297-316.
Skedros JG, Sybrowsky CL, Bloebaum RD, Bachus KN, Wang X (2006c) The relative influence of collagen crosslinks on the mechanical properties of equine cortical bone. In: Transactions of the 52nd Annual Meeting of the Orthopaedic Research Society, p. 1581.
Streeter M, Stout S, Trinkaus E, Burr D (2010) Brief communication: Bone remodeling rates in Pleistocene humans are not slower than the rates observed in modern populations: A reexamination of Abbott et al. (1996). Am J Phys Anthropol, 141, 315-8.
Takano Y, Turner CH, Owan I, et al. (1999) Elastic anisotropy and collagen orientation of osteonal bone are dependent on
the mechanical strain distribution. J Orthop Res, 17, 59-66.
Urbanová P, Novotný V (2005) Distinguishing between human and non-human bones: histometric method for forensic anthropology. Anthropologie, XLIII, 77-85.
van Oers RF, Ruimerman R, van Rietbergen B, Hilbers PA, Huiskes R (2008) Relating osteon diameter to strain. Bone, 43, 476-82.
Verborgt O, Gibson GJ, Schaffler MB (2000) Loss of osteocyte integrity in association with microdamage and bone remodeling after fatigue in vivo. J Bone Min Res, 15, 60-67.
Vincentelli R (1978) Relation between collagen fiber orientation and age of osteon formation in human tibial compact bone. Acta Anat (Basel), 100, 120-128.
Vincentelli R, Evans FG (1971) Relations among mechanical properties, collagen fibers, and calcification in adult human cortical bone. J Biomech, 4, 193-201.
von Ebner V (1887) Über den feineren bau der skeletteile der kalkschwamme usw. Wiener Stizber, 95, 213-236.
Wang XD, Masilamani NS, Mabrey JD, Alder ME, Agrawal CM (1998) Changes in the fracture toughness of bone may not be reflected in its mineral density, porosity, and tensile properties. Bone, 23, 67-72.
Warshaw J (2007) Primate bone microstructural variability: relationships to life history, mechanical adaptation and phylogeny. In Anthropology Ph.D. Thesis), pp. 1-440. New York, USA: The City University of New York.
Williams T, Jardine C, Keenan K, Skedros J, Kiser C (2010) Secondary osteon cross-sectional size and morphotype score are independent in limb bones subject to habitual bending or torsion. Am J Phys Anthropol, Supplement 50, 245.
Yamamoto T, Domon T, Takahashi S, Islam N, Suzuki R (2000) Twisted plywood structure of an alternating lamellar pattern in cellular cementum of human teeth. Anat Embryol (Berl), 202, 25-30.